“The Somni Snore Guard is a truly unique device. Simple to prepare and comfortable to use, it has become a best-seller among serious snoring treatments. We regularly receive unsolicited, positive feedback from satisfied users.”
— British Snoring and Sleep Apnoea Association
CLINICAL TRIALS
British Snoring & Sleep Apnoea Association
According to the British Snoring & Sleep Apnoea Association, an oral vestibular shield encourages natural nasal breathing by gently preventing the mouth from opening during sleep.
The SOMNI Snore Guard is specifically designed for individuals who snore with their mouths open.
Mouth breathing at night can lead to unpleasant side effects such as bad breath (halitosis) and disruptive snoring—both of which affect sleep quality and disturb those around you. By promoting nasal breathing, the SOMNI Snore Guard helps you enjoy fresher breath, quieter nights, and more restful sleep.

Our Values
Efficiency
Comfort
Safety
Trial Overview
UNDERSTANDING SNORING
Snoring is a common condition that affects millions worldwide—more frequently in men than women—and tends to worsen with age. Studies report that between 10% and 45% of adults snore regularly, and the majority of people over 65 are affected.
Habitual snoring is more than just a nuisance. It can cause emotional strain between partners, social embarrassment, and in severe cases, progress to serious health issues such as obstructive sleep apnoea (OSA), high blood pressure, cardiac arrhythmias, and secondary polycythaemia.
Why Snoring Happens
Snoring has multiple causes, including:
- Loss of muscle tone in the throat with age
- Anatomical abnormalities, trauma, or allergies affecting the airway
- Excess body weight
- Sleeping on the back or with the mouth open
- Alcohol consumption before sleep
- Eating large meals shortly before bedtime
These factors can partially collapse or obstruct the airway, creating turbulent airflow past the soft palate, uvula, tongue, and throat tissues. The vibration of these tissues produces the familiar sound of snoring.
Treatment Approaches
Because snoring has many causes, experts recommend a comprehensive approach—from lifestyle changes to mechanical, medicinal, or surgical interventions. Notably, clinical studies have shown high success rates simply by preventing mouth breathing with custom-fitted vestibular mouth shields.
Study Aim
This study set out to evaluate the efficiency of an anti-snoring oral vestibular shield that can be fitted chairside, offering a simple, non-invasive solution for habitual snorers.
STUDY SUMMARY
In a clinical evaluation, stock intra-oral vestibular shields made from heat-malleable, semi-flexible ethylene vinyl-acetate were fitted chairside to the mouths of 77 chronic snorers. Effectiveness was assessed 8–10 weeks after fitting.
- 77.9% of participants benefited from using the device.
- Among those who did not benefit, 11.7% showed signs of nasal or naso-pharyngeal obstruction.
- 6.5% discontinued due to poor self-motivation.
- Only 2 participants were unable to tolerate the shield in the mouth.
These results demonstrate the SOMNI Snore Guard’s strong potential as a safe, effective solution for chronic snoring. Its role in managing obstructive sleep apnoea (OSA) warrants further investigation, highlighting the device’s promise in broader sleep health management.
Results
At the two-week recall, the majority of subjects were progressing well. Two withdrew from the program due to intolerance of the device, while another two required minor adjustments. A common early complaint was loss or unconscious removal of the shield during sleep, which was addressed by reassuring participants of the need for oral adaptation. Those reporting nasal breathing difficulties were advised to use over-the-counter nasal decongestants.
By the conclusion of the clinical trial:
- 66.2% reported complete comfort and complete or near-complete elimination of snoring.
- 11.7% experienced sufficient reduction in snoring frequency or intensity to justify continued use.
- A substantial number described the shield as a “comforter” and reported enjoying sleeping with it.
- Of four subjects with obstructive sleep apnoea, two reported relief from both snoring and OSA symptoms.
- 6.5% found the shield acceptable but continued to snore nasally.
- 5.2% struggled with enforced nasal breathing and frequently expelled the device during sleep.
- 6.5% abandoned the trial due to lack of motivation.
- One subject continued to snore through the mouth despite wearing the shield comfortably.
These findings demonstrate strong overall effectiveness, high comfort levels, and promising potential for broader application in managing snoring and obstructive sleep apnoea.
Discussion
Over 300 anti-snoring devices have been registered with the U.S. Patent Office, yet few have achieved widespread use—suggesting limited effectiveness. One of the greatest challenges in treating snoring is securing the cooperation of the snorer. Any treatment must be non-threatening, comfortable, and ideally improve the quality of life for both the snorer and their sleeping partner. If this is not achieved, even effective solutions are likely to be rejected. Particular care must be taken when introducing a device into the sensitive oral environment.
The fact that only 2 of 77 participants rejected the mouth shield outright speaks strongly to its acceptability. Those who abandoned the trial due to lack of motivation did not reject the device for discomfort, but rather appeared to join the program to appease their partners without genuine commitment to solving their snoring problem.
The oral vestibular shield used in this study was based on earlier work by Campion (1985), who developed a semi-flexible PVC shield molded to dental casts. Over two years, the design was refined to include a palatal tag to prevent dislodgment during normal nocturnal movements. Ventilation holes were added to prevent pressure build-up and allow limited oral airflow, while still encouraging nasal breathing. Importantly, the stock shield could be adapted chairside to individual mouths, making it practical and accessible.
Campion reported 80% improvement among selected subjects, though his exclusion of 39 volunteers reduced the overall effectiveness to 50% when considering the full group. In contrast, this study included all volunteers who could breathe through the nose with the mouth closed, ensuring a more representative sample.
The 77.9% success rate achieved here validates the oral vestibular shield as a first-line treatment option for chronic snoring. For participants who continued to snore nasally or struggled with enforced nasal breathing, the device served as a valuable diagnostic tool, identifying possible nasal or nasopharyngeal obstructions and prompting referral to ENT specialists.
Materials and Methods
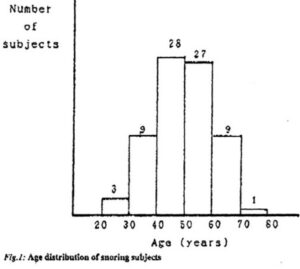
The clinical trials were publicized in a daily newspaper inviting volunteer participants. The first 80 respondents were admitted to the program. One person failed repeatedly to arrive for his appointment while 2 persons withdrew due to ill health, leaving 11 female and 66 male participants. The age distribution of the volunteers is shown in Fig.1, the average age being 49,5 years.

Subjects were counseled in small groups regarding the multi factorial actiology of snoring and informed of the problem that may encounter in adapting to the vestibular shield. In particular, the need to ensure at least one clear nasal passage before retiring to sleep was emphasized. Subjects were also cautioned against unrealistically high expectations and it was stressed, that the oral shield would at best be a means of controlling, rather than curing their snoring. Subjects were interviewed individually and a relevant medical history was recorded. Each subject was required to demonstrate the ability to breathe normally with a closed mouth.
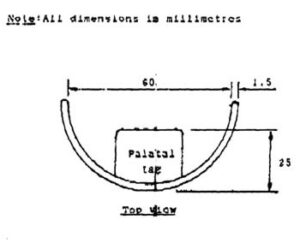
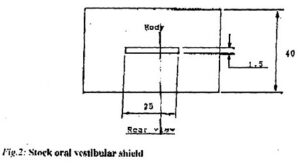
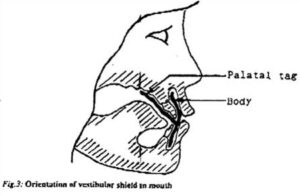
Stock mouth shields, consisting of a curved body and palatal tag (Fig.2) were pre-fabricated from a flexible ethylene vinyl acetate (EVA) material. The stock shield was trimmed to 5mm distal to the second maxillary bicuspid and to 2-3mm short of the limit of the maxillary and mandibular labiobuccal sulci, with the subject in centric occlusion. The shield was placed in water just off the boil (80 – 90ºC) for 10 – 15 seconds. When malleable, the shield was introduced into the subject’s mouth, with the body in the labiobuccal vestibule and the tag clenched lightly between the teeth. The subject was instructed to close the lips and, to simultaneously apply suction and to press the tag against the palate with the tongue for one minute. If necessary, the molding procedure was repeated, until both body and tag were snugly adapted to the subject’s mouth (Fig.3).
Areas of over-extension and interferences with fraena were trimmed with a pair of sharp scissors. Two 2mm diameter holes were punched in the body of the shield in the canine region at the level of the occlusal plane.
Subjects were encouraged to contact the author should they experience difficulties.
The first formal check on progress took place two weeks after the fitting session. The efficacy of the shield was assessed 8 – weeks after the initial fitting. Subjects were questioned regarding the comfort and frequency of use of the shield, while the sleeping partners were asked to report on the effectiveness of the device.
Conclusions
This study has shown, that by wearing an appliance, which prevented mouth breathing during sleep, snoring was controlled in a large proportion of chronic sufferers. The stock oral vestibular shield can be effectively adapted at chair side to the individual and is well tolerated in the mouth. In the vent of possible respiratory distress, the device is readily expelled by the used. The shield could be further viewed as a diagnostic device to identify the snorer who may require more aggressive medical or surgical treatment. The possibility of using the shield for the management of obstructive sleep apnoea warrants further investigation.
Acknowledgements
The author would like to register the contribution of his wife, Alma Veresas observer and co-designer of the oral vestibular shield and to thank his octogenarian for the fabrication of the stock shields. Thanks are also extended to the volunteer snorers for their time and co-operation during the clinical trial.
References
Anderson, L & Brattstrom, V (1991) Cephalometric analysis of permanently snoring patients with and without obstructive sleep apnoea. The International Journal or Oral and Maxillofacial Surgery, 20, 159-162.
Block, AJ, Boysen, PG. Wynne, JW & Hunt, LA (1979) Sleep apnoca, hypapnoen and oxygen desaturation subjects. New England Journal of Medicine, 300, 513-517.
Campion, PCS (1985) The management of snoring: Background and a series of treated cases. Medical Journal Of Australia, 143, 337-338.
Fujita, AS, Conway, W, Zorick, F & Roth, T (1981) Surgical correction of anatomic abnormalities in obstructive sleep apnoea syndrome: Uvulopalate-pharyngoplasty, Otalaryngology – Head and Neck Surgery, 89, 923-024.
Lipman, DS (1990) Stop your husdband from snoring, 1st ed, Ch.8, pp.118-152, Emmanus, Pennsylvania: Rondale Press.
Rice, OH & Persky, M (1986) Snoring: Clinical applications and treatment. Otolaryngoly, Head and Neck Surgery, 95, 28-30.
Wagner, G & Price, RR (1987) An intraoral device to pravent snoring, General Dentistry, May/June, 212-213.
EFFECTIVE ANSWER TO YOUR SNORING PROBLEMS
Why SOMNI snore guard? My story...
As a sufferer of snoring for many years I searched for a simple non-surgical solution to my snoring problem that irritated my wife and affected my marriage.
Eventually after many years and many product failures (pillows, drops, oil rubs) I was considering a surgical solution, besides the cost I was concerned of the permanent effect, and the long term trauma that my body would endure in the operation… It so happened that I had a routine dental appointment a week before the surgery… this was to change my life forever.
You see unbeknown to me my dentist (Dr Veres) had developed the SOMNI Snore guard, once revealing my condition and frustration an intense discussion followed, naturally I had a hesitant approach to a quick fix solution… I simply did not believe that a simple mouth guard would solve my snoring, after all I have been down this road and lost… the persisted plea from the doctor and the constant nagging from my wife encouraged me to prove them wrong!
That night angrily and with great determination to prove them wrong… I followed the instructions and inserted the snore guard and went to bed… What a shocker!
You see every night, all night I would normally toss and turned as the snoring kept me awake, but the morning after using the Somni snore guard I felt great, rested, energized and ready to face the day ahead… but did the snoring stop?
I ran to the kitchen to confront my wife, she was whistling whilst preparing breakfast and yes… she confirmed not a sound! … and added “I told you so”!
I was so impressed; I wanted to share this with the world… I promptly acquired the rights to sell the product online!
We are happy to announce that we have helped thousands of people with their snoring problem; you too can improve the quality of your life forever!
In order to help as many people as possible to overcome their snoring problem we have reduced the pricing… if you’re interested, act quickly, the pricing will revert back to normal shortly!





